FDA Approved Papzimeos: First Immunotherapy for Recurrent Respiratory Papillomatosis
Historic FDA Approval for RRP
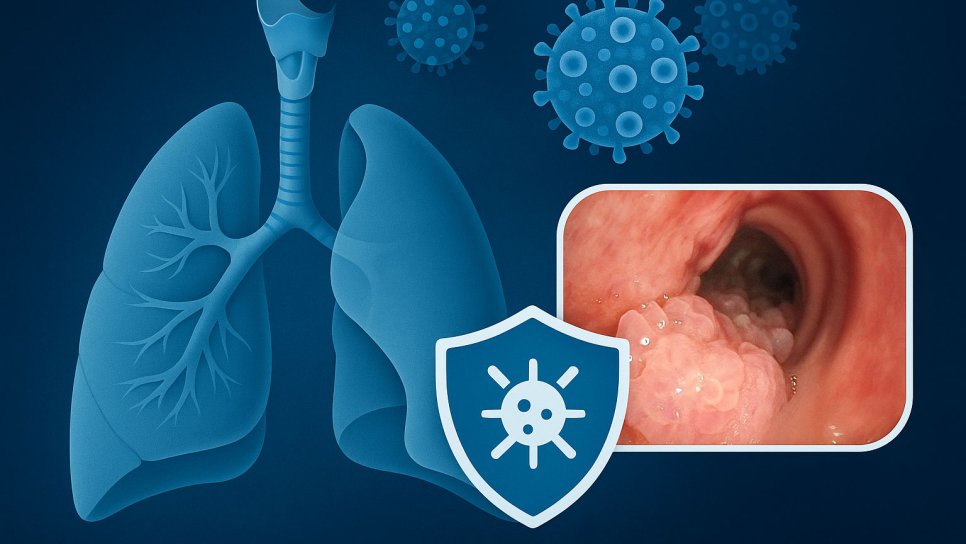
Recurrent respiratory papillomatosis (RRP) is a rare but serious disease caused by HPV types 6 and 11. Benign tumors develop in the larynx and airways, but these tumors recur persistently. Patients often require repeated surgeries, which both reduce quality of life and cause serious problems with voice and breathing. Until now, there has been no approved medical therapy for RRP.
💡 On August 14, 2025, the FDA announced the approval of a new immunotherapy called Papzimeos. This represents a historic milestone in the treatment of this disease.
🫁 What is Recurrent Respiratory Papillomatosis (RRP)?
RRP is a rare disease caused by HPV types 6 and 11, in which wart-like benign tumors develop in the airways. It most commonly affects the vocal cords (larynx), leading to symptoms such as hoarseness, shortness of breath, and chronic cough. In children, these rapidly growing tumors can cause severe breathing problems during sleep.
- Responsible HPV types: HPV 6 and 11
- Mode of transmission: Sexual contact or vertical transmission from mother to child during birth
- Prevalence: Approximately 2 cases per 100,000 children
- Treatment: Currently, the main option is surgery; however, due to frequent recurrences, patients may require multiple surgeries each year
💊 What is Papzimeos?
- Active substance: Zopapogene imadenovec-drba
- Mechanism: Non-replicating adenoviral vector-based immunotherapy
- Administration: Subcutaneous injection
- Target: To activate the immune system against cells infected with HPV 6 and 11
- 📌 Marketing authorization holder: Precigen, Inc.
➡️ Unlike current treatment approaches that rely solely on surgery, this represents the first option designed to harness the immune system.

📊 Clinical Trial Data
The approval of Papzimeos (investigational name PRGN-2012) is based on the phase 1/2 pivotal trial published in The Lancet Respiratory Medicine in April 2025 (DOI:10.1016/S2213-2600(24)00368-0).
This was a single-center, single-arm, open-label study. Participants were adult RRP patients who had required at least three surgical interventions in the previous year. All patients received four subcutaneous injections after surgical debulking (on days 1, 15, 43, and 85).
- Number of patients: 38 patients were enrolled between March 2021 and June 2023; 35 of them were treated at the recommended phase 2 dose of 5×1011 viral particles.
- Primary endpoint: Complete response defined as no need for surgery for 12 months.
Findings
- 51% (18/35) complete response: Half of the patients required no additional surgery for 12 months (95% CI 34–69).
- Duration of response: Median duration has not yet been reached; most responses were maintained for up to 2 years.
- Immune response: Strong correlation observed between clinical benefit and activation of HPV 6/11-specific T cells.
- Safety: Adverse events were mostly mild to moderate. Most common were:
- 97% injection site reactions
- 80% fatigue
- 71% chills
- 69% fever
Conclusion: PRGN-2012 eliminated the need for surgery for 12 months in 51% of adult RRP patients and was found to be safe. Based on these findings, a biologics license application was submitted to the FDA, and in August 2025, it was approved under the brand name Papzimeos.
🌍 What Does This Approval Mean?
- Approximately 1,000 new RRP cases are diagnosed each year in the United States.
- Although rare, the disease has devastating effects on patients’ quality of life.
- With this approval, a new era begins for patients who no longer have to rely on repeated surgeries.
💬 FDA official Dr. Vijay Kumar: “This approval has the potential to transform the treatment landscape for RRP and provide lasting relief to patients who are dependent on recurrent surgeries.”
✅ In summary: Papzimeos has become the first immunotherapy approved by the FDA for RRP. It eliminated the need for surgery in half of patients and demonstrated durable efficacy. This is considered a groundbreaking development in the field of rare diseases.
FDA Approves First Immunotherapy for Recurrent Respiratory Papillomatosis. For Immediate Release: August 14, 2025. https://www.fda.gov/news-events/press-announcements/fda-approves-first-immunotherapy-recurrent-respiratory-papillomatosis






