
Revolutionizing Mitochondrial Disease Prevention: The Three-Parent Baby Technology
Mitochondrial diseases are often fatal inherited disorders that affect organs with high energy demands, such as the nervous system, muscles, and heart.
New technologies – mitochondrial donation and preimplantation genetic testing – offer promising solutions to prevent transmission of these diseases to children.
📅 Publication Details and Scientific Context
This article is based on data from three separate scientific documents simultaneously published in The New England Journal of Medicine (NEJM) on July 16, 2025:
- Original Research: Mitochondrial Donation and Preimplantation Genetic Testing for mtDNA Disease
- Brief Clinical Report: Mitochondrial Donation in a Reproductive Care Pathway for mtDNA Disease
- Editorial Review: Advances in Preventing Transmission of Mitochondrial DNA Diseases
💉 Mitochondrial Diseases: Definitions, Examples, and Prevalence
Mitochondrial diseases are inherited metabolic disorders that impair the mitochondria’s ability to produce energy. They typically result from mutations in the mitochondrial DNA (mtDNA) passed down from the mother.
Most Well-Known Mitochondrial Diseases:
- Leigh Syndrome
- MELAS (Mitochondrial Encephalomyopathy, Lactic Acidosis, and Stroke-like Episodes)
- LHON (Leber’s Hereditary Optic Neuropathy)
- MERRF (Myoclonic Epilepsy with Ragged Red Fibers)
Prevalence:
- Approximately 1 in 4,300 individuals is affected by a mitochondrial disease.
- 1 in every 200 people may carry a pathogenic mtDNA variant.
Common Symptoms:
- Muscle weakness and exercise intolerance
- Cardiomyopathy
- Vision and hearing loss
- Seizures
- Developmental delays
- Encephalopathy and stroke-like episodes
The severity of symptoms is associated with the level of heteroplasmy.
🧬 What is Mitochondrial Donation?
Mitochondrial donation involves transferring the nucleus from a fertilized egg of a woman with diseased mitochondria into a donor egg that has had its own nucleus removed.
- It is suitable for homoplasmic women (those with 100% affected mtDNA).
- It is also preferred in cases with a high level of heteroplasmy.
⚖️ Scope and Design of the Scientific Study
This comparative observational study aimed to evaluate the real-world outcomes of two assisted reproductive technologies in women who carry mitochondrial disease.
A total of 61 women carrying pathogenic mitochondrial DNA (mtDNA) variants were enrolled. Based on their mutation load (heteroplasmy level) and personal preferences, they were divided into two groups:
-
22 women received Pronuclear Transfer mitochondrial donation.
-
39 women underwent Preimplantation Genetic Testing (PGT).
What is PGT?
Preimplantation Genetic Testing (PGT) involves removing a few cells from embryos created via conventional IVF and performing genetic analysis. In women carrying mtDNA mutations, the mutant mitochondrial load (heteroplasmy) of each embryo is measured. Only low-risk embryos are transferred to prevent disease transmission.
Importance of Comparative Design: Two different approaches applied to patients with similar clinical risks were evaluated prospectively. This allowed for a direct comparison of clinical success rates and mtDNA levels in offspring.
This research revealed the strengths of pronuclear transfer and PGT in terms of both efficacy and safety, offering practical clinical guidance on which strategy may be more suitable for different patient groups.
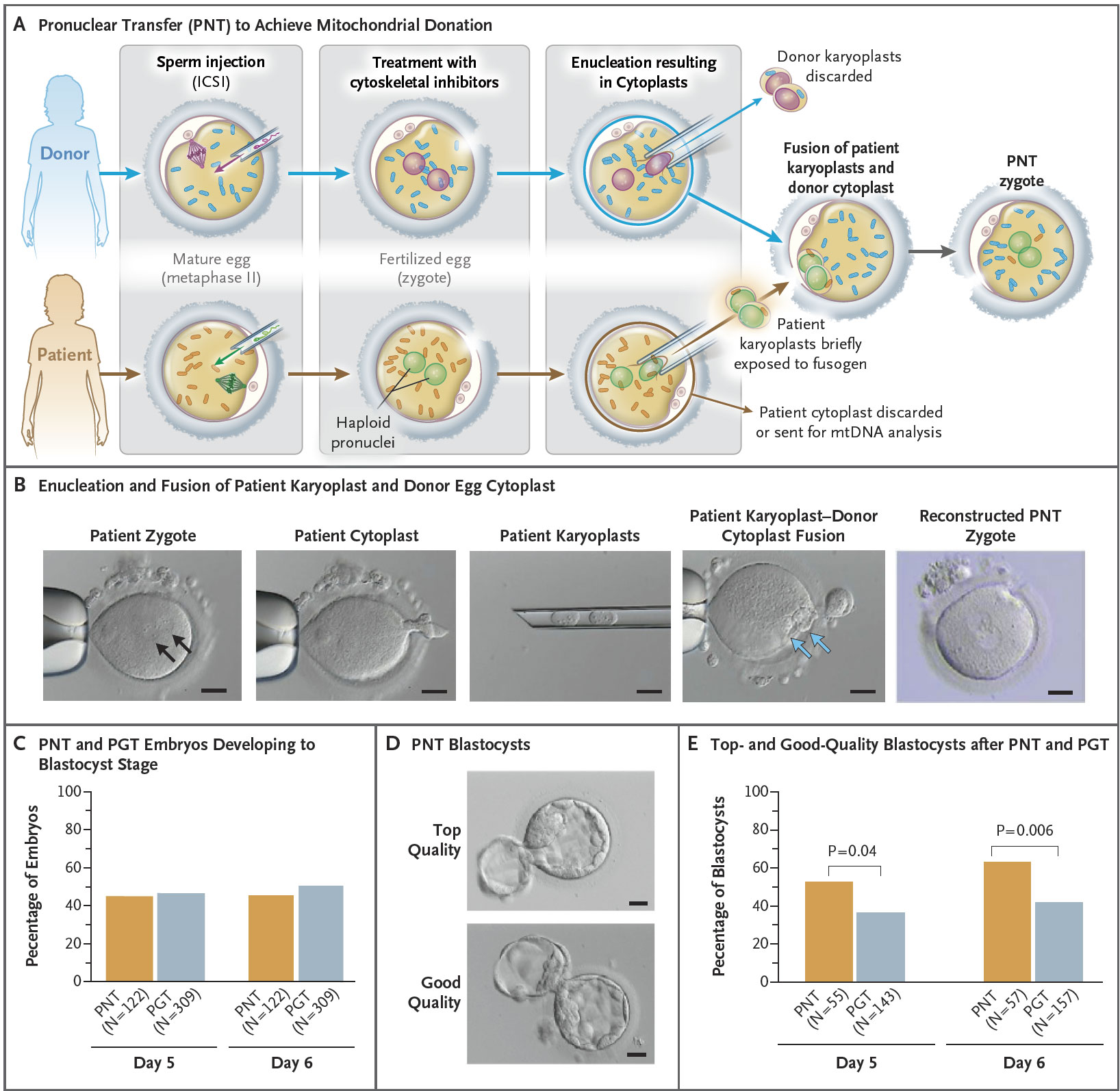
Figure 1. Pronuclear-Transfer Procedure and Comparison of Embryo Development with Pronuclear Transfer and with PGT.
Panel A shows the pronuclear-transfer procedure. The images in Panel B show the enucleation of patient karyoplasts and the fusion of patient karyoplasts with the donor egg cytoplast (scale bar, 20 μm). Arrows in the first image indicate the pronuclei, and arrows in the fourth image indicate the karyoplasts. Panel C shows a comparison of the percentage of embryos with blastocyst formation on days 5 and 6 after intracytoplasmic sperm injection in the pronuclear-transfer group and in the PGT group. The images in panel D show examples of top-quality and good-quality pronuclear-transfer blastocysts (scale bar, 20 μm). Panel E shows the percentage of blastocysts that were top quality or good quality after pronuclear transfer and after PGT on days 5 and 6. The percentage was higher in the pronuclear-transfer group than the PGT group on both day 5 (P=0.04 by chi-square test) and day 6 (P=0.006 by chi-square test).
📊 Study Data
| Method | Pregnancy Rate | Live Births |
|---|---|---|
| Pronuclear Transfer | 36% (8/22) | 8 + 1 ongoing |
| PGT | 41% (16/39) | 18 |
🧬 mtDNA (Heteroplasmy) Levels in Newborns
Genetic analysis of blood samples from newborns revealed the proportion of pathogenic variants in mitochondrial DNA (% heteroplasmy). This level is a critical marker of the child's disease risk.
- In babies born via Pronuclear Transfer:
- Heteroplasmy levels ranged from 0% to 16%.
- In 6 babies, the level of mutated mtDNA was reduced by 95% to 100%.
- In 2 babies, the reduction was between 77% and 88%.
- In babies born via PGT: Heteroplasmy levels ranged from 0% to 7%.
Note: Medically, heteroplasmy levels below 15% are generally considered non-pathogenic and safe.
👶 Clinical Follow-up and Health Outcomes
Newborns were regularly monitored for health status after birth. The findings are as follows:
- All babies were assessed as healthy at birth.
- Only 2 babies showed mild and temporary side effects:
- One baby had infantile myoclonic epilepsy (resolved spontaneously without requiring treatment).
- The other had a congenital heart rhythm disorder (associated with hyperlipidemia); it responded completely to medical therapy.
⚠️ Ethical and Legal Debates
- The concept of "three-parent baby" raises ethical concerns due to the embryo receiving genetic material from three individuals (mother, father, and mitochondrial donor).
- Pronuclear transfer involves advanced genetic manipulation of human embryos and is therefore subject to limited approval in countries such as the United States.
- This technology is only applied to women carrying pathogenic mtDNA and requires ethical board approval.
The United Kingdom was the first country to officially authorize this technology and integrate it into clinical practice through the NHS-supported center in Newcastle.
This practice is not yet legal in Türkiye. However, in light of growing scientific evidence, ethical discussions on its adoption seem inevitable.
📌 Conclusion
The comprehensive dataset published in NEJM 2025 supports the clinical success and safety of two advanced technologies that may halt the intergenerational transmission of mitochondrial diseases.
Mitochondrial donation and PGT are no longer merely experimental—they have the potential to become part of future standard clinical practice.
As science pushes boundaries, ethical frameworks must also evolve to keep pace with innovation.
Hyslop LA, Blakely EL, Aushev M, Marley J, Takeda Y, Pyle A, Moody E, et al. Mitochondrial donation and preimplantation genetic testing for mtDNA disease. N Engl J Med. 2025 Jul 16; DOI: 10.1056/NEJMoa2415539.






