Y-90 Resin Microspheres: FDA Approval Broadens Therapeutic Scope in Unresectable HCC
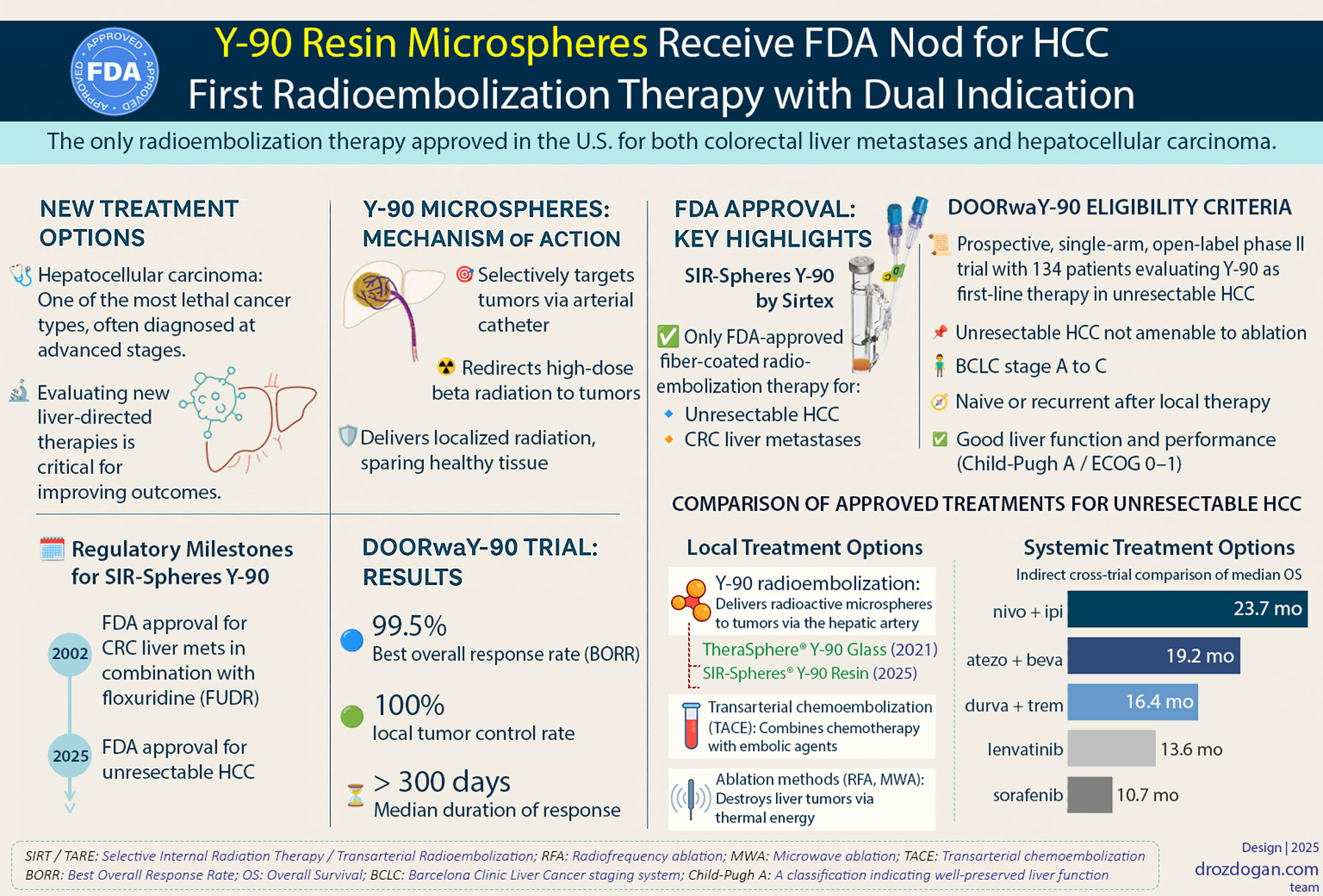
With this approval, these microspheres become the only radioembolization therapy approved for both colorectal cancer liver metastases and HCC in the United States.
Radioembolization—commonly referred to as selective internal radiation therapy (SIRT)—uses personalized dosimetry to deliver the optimal radiation dose directly to tumors in patients with HCC.
This approval offers clinicians expanded flexibility in selecting a liver-directed therapy tailored to individual patient needs and treatment goals.
Hepatocellular carcinoma (HCC), commonly known as liver cancer, ranks among the deadliest cancers globally and is unfortunately often diagnosed at advanced stages. For patients whose tumors cannot be surgically removed (unresectable), improving quality of life and prolonging survival remains one of oncology’s most pressing challenges. Addressing this critical need, the recent FDA approval of Y-90 resin microspheres represents a groundbreaking development in targeted liver cancer therapy.
This article explores the significance of the FDA's approval, the scientific mechanism and evidence supporting Y-90 microspheres, patient eligibility criteria, and comparisons with other approved treatment options, including survival data.
What Are Y-90 Resin Microspheres? A Personalized Approach to Tumor Targeting
Y-90 resin microspheres, also known as Selective Internal Radiation Therapy (SIRT) or Transarterial Radioembolization (TARE), deliver localized radiation therapy directly to liver tumors. These microscopic spheres (20-60 micrometers in diameter), smaller than a human hair, are embedded with Yttrium-90, a radioactive isotope.
During the procedure, interventional radiologists access the femoral artery and guide a thin catheter to the hepatic artery supplying the tumor. Due to the tumor’s high vascularity, Y-90-loaded microspheres preferentially accumulate in tumor tissue, delivering high-dose beta radiation while minimizing exposure to surrounding healthy liver tissue. This personalized dosimetry approach calculates the optimal radiation dose based on each patient's tumor load, liver function, and vascular characteristics, significantly enhancing both efficacy and safety.
FDA Approval: A Dual Milestone in Liver Cancer Treatment
The U.S. Food and Drug Administration (FDA) approved Sirtex Medical’s SIR-Spheres Y-90 resin microspheres for treating unresectable hepatocellular carcinoma. Notably, this approval makes Y-90 microspheres the first and only radioembolization therapy approved by the FDA for both liver metastases from colorectal cancer and unresectable HCC. This dual indication underscores the broad efficacy and robust safety profile of this innovative therapy across diverse patient groups.
DOORwaY-90 Trial: Strong Scientific Evidence and Remarkable Results
The FDA’s decision was based on promising results from the prospective, multicenter, open-label DOORwaY-90 trial, involving 100 patients across 18 U.S. centers. This study meticulously evaluated the effectiveness and safety of Y-90 microspheres in unresectable HCC patients who had not previously received systemic therapy.
Key patient eligibility criteria included:
-
Unresectable tumors: Tumors that could not be completely removed surgically.
-
Ineligibility for other ablative treatments: Not suitable for radiofrequency ablation (RFA) or microwave ablation (MWA).
-
BCLC stages A, B1, B2, or selected C: Including cases with limited portal vein thrombosis or minimal extrahepatic spread.
-
Good liver function and general health: Child-Pugh A5/A6 and ECOG performance status 0–1.
-
Tumor size constraints: Maximum single tumor diameter <8 cm; total tumor diameter <12 cm.
98.5%: Best overall response rate
100%: Local tumor control rate
Median duration of response > 300 days
Dr. Armeen Mahvash, co-principal investigator of DOORwaY-90, emphasized, "This study advances the field of radioembolization, providing reproducible dosimetry outcomes and a strong safety profile, leading to very promising clinical results."
Who Benefits Most from Y-90 Resin Microspheres?
SIR-Spheres are particularly beneficial for patients with unresectable HCC who have:
-
No macrovascular invasion.
-
Child-Pugh A cirrhosis and well-compensated liver function.
-
Good performance status (ECOG 0–1).
-
Limited or no extrahepatic disease.
It is important to clarify that Y-90 microspheres constitute a local treatment approach. They specifically target liver tumors and are ideal for patients with minimal or no distant metastasis, focusing on controlling the liver tumor burden rather than addressing systemic spread.
Other Approved Treatment Options for HCC and Survival Outcomes
While the FDA approval of Y-90 microspheres marks significant progress, several other FDA-approved systemic and local treatments are also available for unresectable HCC:
Systemic Therapies:
-
Nivolumab + Ipilimumab: Median Overall Survival (OS) ~23.7 months.
-
Atezolizumab + Bevacizumab: Median OS ~19.2 months.
-
Durvalumab + Tremelimumab (STRIDE): Median OS ~16.4 months.
-
Lenvatinib: Median OS ~13.6 months.
-
Sorafenib: Median OS ~10.7–13.4 months.
-
Cabozantinib, Regorafenib, Ramucirumab: Median OS ~9–10 months.
Local Therapies:
-
Transarterial Chemoembolization (TACE): Median OS ~20–30 months; PFS can extend to ~10.3 months when combined with immunotherapy.
-
RFA/MWA: 5-year OS of approximately 50–70%, with local recurrence-free survival of ~1–3 years.
Conclusion: The Future of Personalized Liver Cancer Treatment
The FDA approval of Y-90 resin microspheres underscores the importance and potential of personalized treatments in liver cancer care. Offering high local tumor control, extended response durations, and an exceptional safety profile, Y-90 microspheres emerge as a crucial option in treating unresectable HCC. As we move forward, this innovative therapy promises to play a pivotal role within multidisciplinary treatment frameworks, potentially combined with other modalities to optimize patient outcomes.
This advancement represents not just a new therapeutic option but also hope for many patients facing the challenging reality of liver cancer.