How Survival Rates Changed in Urothelial Cancer Before and After Immunotherapy and ADCs
Real-World Data on Immune Checkpoint Inhibitors and Antibody-Drug Conjugates
🔍 Why Is It Important?
- Urothelial cancers include tumors arising from the bladder, ureters, renal pelvis, and urethra; more than 90% of these originate from the bladder.
- Globally, about 573,000 new bladder cancer cases are diagnosed each year, with nearly 213,000 deaths from the disease.
- Bladder cancer is one of the top 10 most common cancers in men and is known for its high mortality rate and tendency to recur.
Approximately 10–15% of bladder cancer patients are diagnosed at the metastatic (stage IV) stage. However, a significant proportion of patients initially diagnosed at localized or non–muscle-invasive stages may later develop metastases—especially in muscle-invasive bladder cancer, where this rate may reach up to 50%. Overall, about 30–40% of bladder cancer patients will experience distant metastasis at some point in their disease course.
Metastatic urothelial carcinoma (mUC), including bladder cancer, is generally a group of diseases with a poor prognosis. While traditional chemotherapy yields a 5-year survival rate below 5%, newer therapies targeting immune checkpoints and antibody-drug conjugates (ADCs) have shown potential to change this outlook. However, outside of clinical trial data, their real-world impact has not been fully clear.
📊 A large retrospective analysis published in JAMA Network Open on July 9, 2025, provides a clear answer: New-generation immunotherapies and ADCs significantly improve overall survival.
Graphic Summary
🔬 Study Details
- Data Source: Flatiron Health Electronic Health Record database (USA-based)
- Time Frame: 8,955 patients who started treatment between 2011–2023
- Follow-Up Duration: Through September 30, 2024
- Treatment Eras Included:
- Pre-immunotherapy era (2011–2016) – Chemotherapy-dominated era
- Post-immunotherapy, pre-ADC era (2017–2019) – Introduction of immunotherapies
- Post-ADC era (2020–2023) – Modern era with ADCs in routine use
❗ Patients receiving clinical trial drugs or no treatment at all were excluded from the study.
NOTE: The main ADC used in urothelial cancer is enfortumab vedotin, targeting Nectin-4 (brand name: Padcev). Additionally, sacituzumab govitecan (Trodelvy), targeting Trop-2, is used in selected cases.
📈 Survival Data
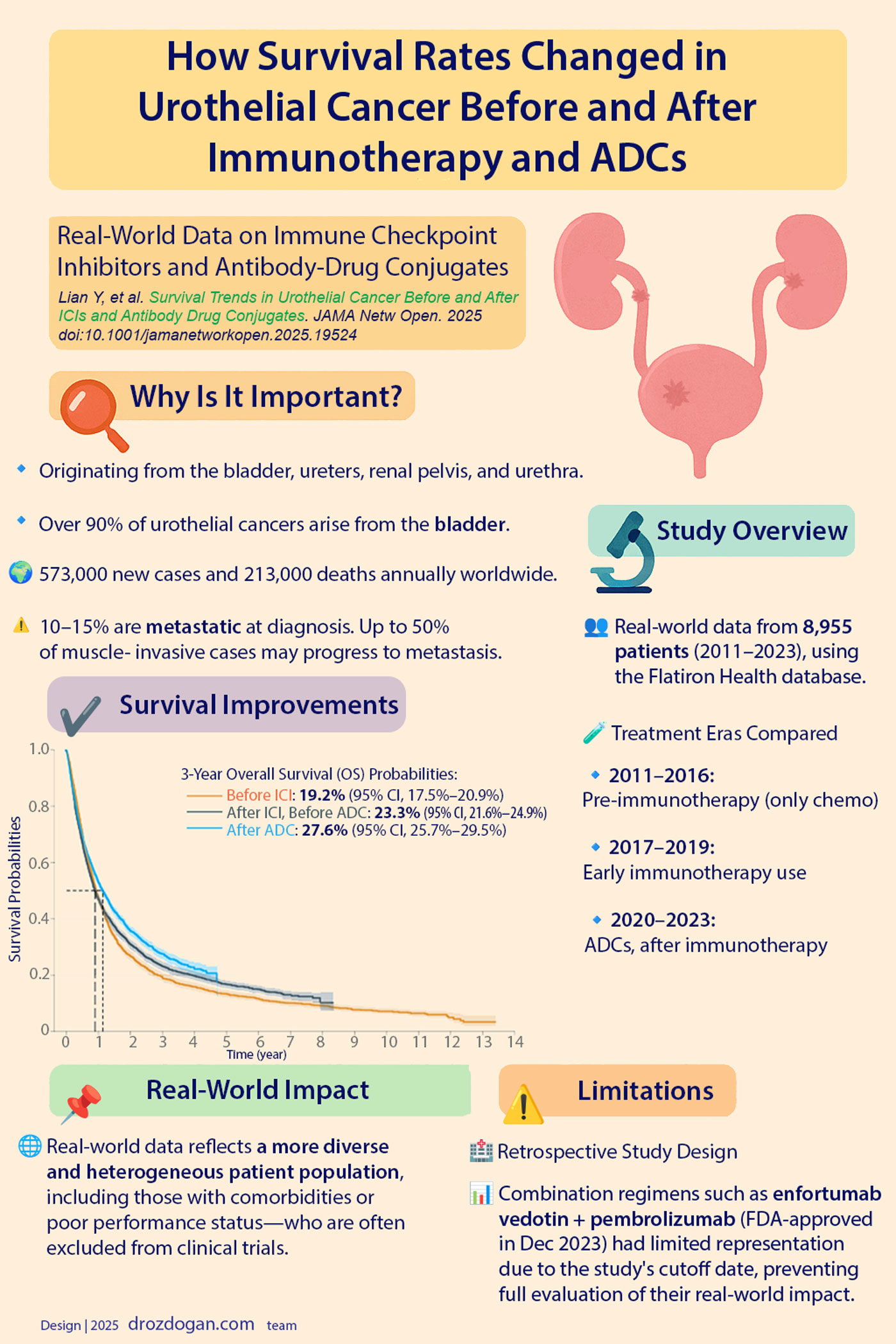
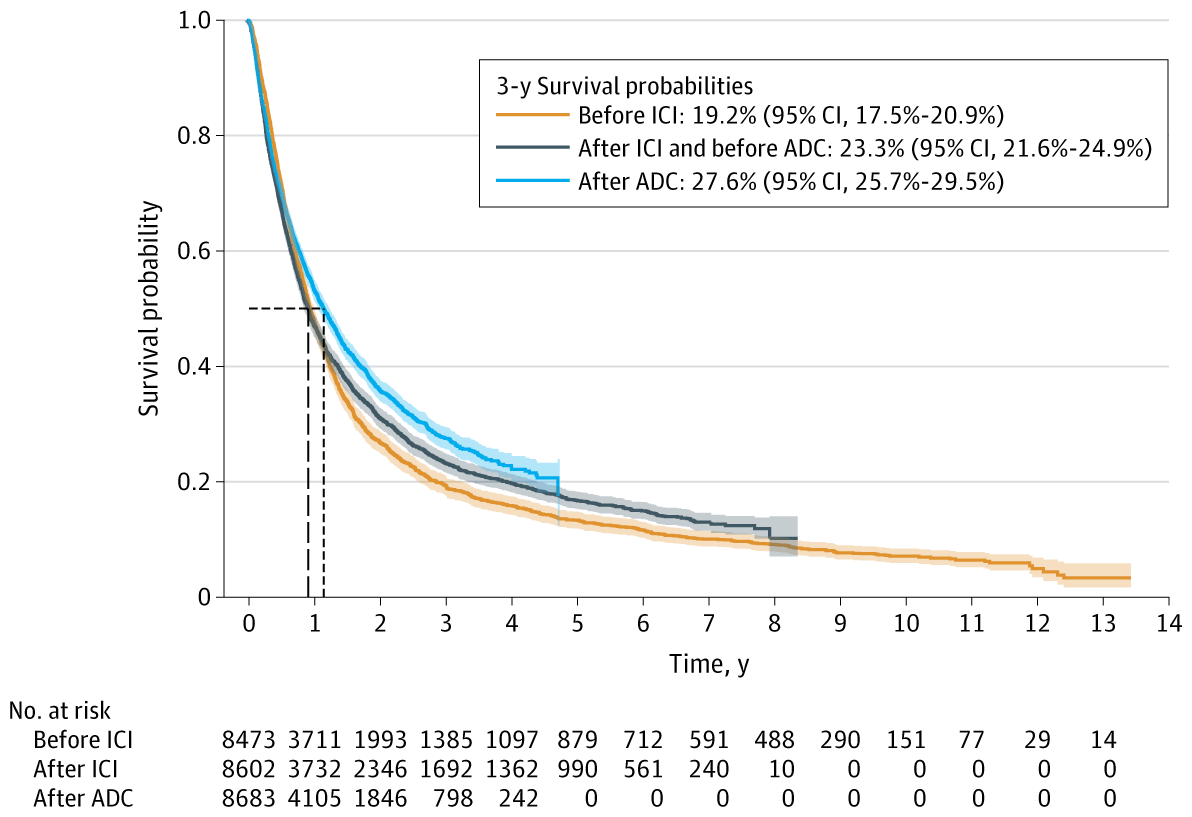
✅ 3-Year Overall Survival Rates:
| Era | 3-Year OS | Confidence Interval |
|---|---|---|
| Pre-immunotherapy (2011–2016) | 19.2% | (17.5%–20.9%) |
| Post-immunotherapy, pre-ADC (2017–2019) | 23.3% | (21.6%–24.9%) |
| Post-ADC (2020–2023) | 27.6% | (25.7%–29.5%) |
🔹 These results correspond to an absolute increase of 8.4% and a relative improvement of 44%.
🕒 Median Overall Survival:
- 2011–2016: 11.1 months
- 2020–2023: 13.6 months (Significant difference, p < 0.001)
📌 Significance of the Study
This study demonstrates that new-generation therapies provide significant survival advantages not only in clinical trials but also in real-world settings for patients with metastatic urothelial cancer.
Specifically:
- The contribution of ADCs such as enfortumab vedotin is clearly evident.
- Future real-world data will likely clarify the impact of ADC + immunotherapy combinations, such as Padcev + Keytruda.
⚠️ Limitations
- The Flatiron database primarily represents community-based clinics, with limited data from academic centers.
- The full impact of ADC + immunotherapy combinations (e.g., enfortumab + pembrolizumab, FDA-approved in December 2023) was not yet assessable in this study.
- As a retrospective study, differences in patient characteristics that may influence treatment outcomes could not be fully eliminated; however, these were minimized as much as possible using statistical techniques (particularly inverse probability weighting).
💬 Conclusion
This large cohort study clearly shows that the improvements in survival observed over the past decade in metastatic urothelial cancer are directly associated with the introduction of immunotherapy and ADCs.
In the long run, these developments will shape both everyday clinical practice and the design of clinical trials for novel treatment combinations.
Lian Y, Voruganti T, Lu J, Long Q, Mamtani R. Survival Trends in Urothelial Cancer Before and After ICIs and Antibody Drug Conjugates. JAMA Netw Open. 2025;8(7):e2519524. doi:10.1001/jamanetworkopen.2025.19524